Mission
- Construct stable, non-toxic, targeted nanomaterials-based therapeutic agents that can be used to treat devastating and debilitating diseases, including cancers
- Develop nanosystems capable of the rapid, inexpensive, accurate, sensitive, and selective detection of biomolecular targets at the point-of-care and in unconventional environments (e.g., inside cells)
- Replace conventionally used diagnostic probes, imaging agents, and therapeutics with nanotechnology-based ones that have the potential to revolutionize the medical field by outperforming the state-of-the-art
- Synthesis nanoparticles that can ramp up the immune system to fight disease or turn it down in the case of an overblown immune response with high specificity
- Develop nanostructured materials that can be used as substrates for cellular growth to understand, and ultimately control, cellular processes
Vision
- Enable the earlier detection of disease so that patient prognoses can be improved
- Improve patient care and save lives by finding more effective treatments
- Eliminate the adverse side-effects caused by standard treatment options
- Provide avenues to personalized and precision medicine, where treatment options can be tailored to the individual based on their genetic code
Active Centers
Spherical Nucleic Acids: a revolutionary new class of therapeutics
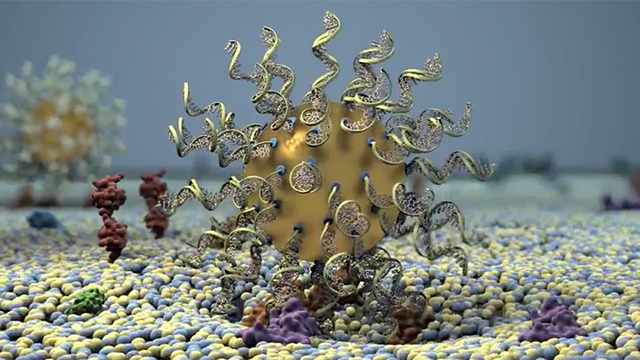
Invented by Chad Mirkin, spherical nucleic acids (SNAs) are typically composed of a nanoparticle core and densely packed and highly oriented nucleic acid shell.
These nanostructures are capable of crossing biological barriers to deliver therapeutics or stimulate an immune response.
This makes SNAs uniquely effective in areas like dermatology, where they can be actively taken up by the skin to target disease-causing genes, and in vaccine design, where they can be used to train cells to fight cancer.
Learn more:
Targeting cancer with gold nanostars

Targeted drug-delivery systems hold significant promise for treating cancer effectively by sparing healthy surrounding tissues. But the promising approach can only work if the drug hits its target.
IIN researchers have developed a new way to determine whether or not single drug-delivery nanoparticles will successfully hit their intended targets. By studying drug-loaded gold nanostars on cancer cell membranes, the researchers found that nanostars designed to target cancer biomarkers transited over larger areas and rotated much faster than their non-targeting counterparts.
By delivering drugs directly into the diseased area — instead of blasting the whole body with treatment — targeted delivery systems result in fewer side effects than current treatment methods.
Learn more: